|
12/22/2023 0 Comments Thermodynamics calculator physics 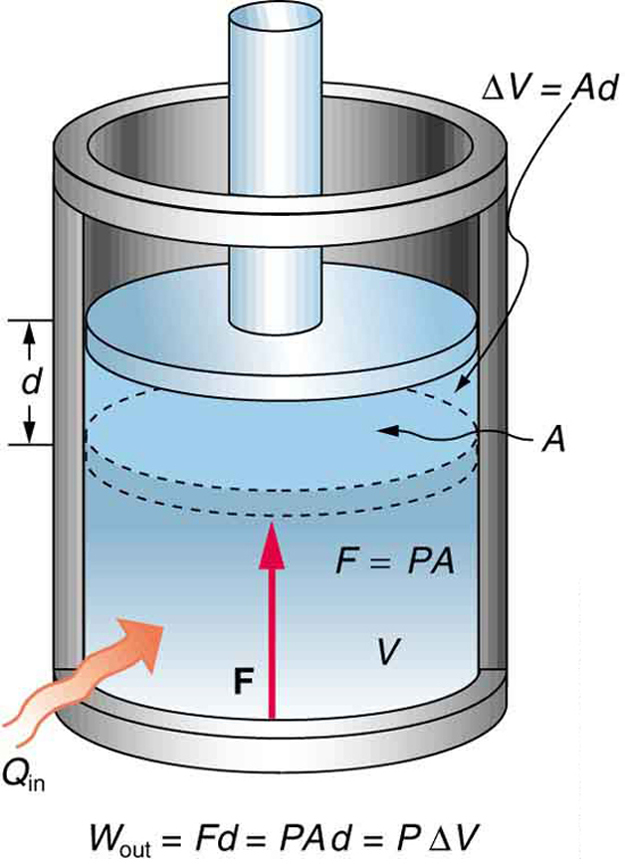
The idea is to load only two scripts at once: the utility module and the script of interest. Equations for the condensation part of convection.Equations for the boiling part of convection.Equations for convection (implicitly only free and forced convection).Linear interpolation of thermophysical properties of air (hard-coded data).Linear interpolation of thermophysical properties of water (hard-coded data).Utility functions: used by every other script.To avoid loading too much instructions at once in the calculator, we separate the formulas in sections: So we must reduce the amount of code as much as possible by designing an efficent wrapping of mathematical formulas. The calculator has no file explorer, i.e. Would make the calculation of such formulas easy. 
The formula is quiet cumbersome to type in a calculator:ĭesigning a script running in the Numworks environment and that prompts the user for input variables Incropera.įor instance, the Churchill-Bernstein equationįor convective heat transfer is used to approximate the dimensionless Nusselt number and thus estimate the averageĬonvection coefficient between two fluids in cross-flow around a cylinder. In particular, the background theory is based on EPFL course ME-341.įor theoretical details, refer to Fundamentals of heat and mass transfer. Of Heat and Mass transfer problems when solving them with paper, a pen and a calculator. This project was motivated by the need to quickly perform redundant calculations 
The usage instructions for different scenarios are listed below the calculator.Implementation of Physics formulas and a convenient user interface for NumWorks calculator. To use the calculator, you need to correctly fill out the table describing interacting substances. This allows a more broad range of problems to be solved. Also, the calculator can take into account the quantity of heat gained or lost to the surroundings. 
This is the equation used by the calculator to find the unknown value. Tn) should be the same, because of thermal equilibrium. Note that the final temperature for all substances (T1, T2. If we replace heat quantities with the formula described here: Quantity of heat, we will get the following equation: That is, the algebraic sum of all heat quantities (gained and lost) in a thermally isolated system equals zero. Where n – number of objects in the system. This is our thermal equilibrium equation. In the case of a thermally isolated system, we can say that during an interaction between objects inside a system (until it reaches thermal equilibrium), the amount of energy gained by one object must be exactly equal to the amount of energy lost by another. The first law of thermodynamics can be stated as follows: during an interaction between a system and its surroundings, the amount of energy gained by the system must be exactly equal to the amount of energy lost by the surroundings. In physics, the law of conservation of energy states that the total energy of an isolated system in a given frame of reference remains constant - it is said to be conserved over time. The thermodynamic system is called a thermally isolated system if it does not exchange mass or heat energy with its environment. A system is said to be in thermal equilibrium with itself if the temperature within the system is spatially and temporally uniform. This is called the zeroth law of thermodynamics. Two objects are in thermal equilibrium if no heat flows between them when they are connected by a path permeable to heat, that is, they both have the same temperature. In the process of reaching thermodynamic equilibrium, heat is transferred from the warmer to the cooler object. To solve the problem, it uses the thermal equilibrium equation, more on this below. The only condition is that there should not be any phase transition (or phase change) of substances. This online calculator can solve thermodynamic equilibrium problems, such as finding the final temperature when mixing fluids, or finding the required temperature for one of the fluids to achieve a final mixed temperature. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
